The quest to find effective treatments for the more than 10 million Americans who suffer from autoimmune diseases has spanned decades. Autoimmune diseases—which include rheumatoid arthritis (RA), psoriatic arthritis, Crohn’s disease, and ulcerative colitis—are a family of very debilitating diseases in which a patients’ immune system attacks their own, healthy tissues and organs. These diseases, if untreated, lead to irreparable damage to those tissues.
While there have been significant advancements in the treatment of autoimmune disease over the past two decades, the current state-of-the-art therapies—namely, a group of immunosuppressive drugs, often called “biologics,” which work by “turning off” or “blocking” the immune system—have numerous drawbacks and very high costs. As such, there are significant opportunities for alternative solutions.
SetPoint Medical has developed such an alternative that could potentially downregulate inflammation without causing immunosuppression. The company has developed an innovative approach that uses a small implantable device rather than pharmaceuticals to treat autoimmune diseases.
The company has developed an innovative approach that uses a small implantable device rather than pharmaceuticals to treat autoimmune diseases.
Today, Norwest Venture Partners announced that it recently led an $80 million preferred stock financing for SetPoint. In addition, the company announced a senior secured term loan facility of up to $65 million from Runway Growth Capital. These financings will enable SetPoint to complete its ongoing pivotal trial for the company’s initial indication, Rheumatoid Arthritis, file for regulatory approval with the Food and Drug Administration (FDA), and begin commercialization in the U.S.
To appreciate why we are excited about SetPoint’s approach, it is important to understand some of the problems with the biologic therapies for autoimmune diseases and how SetPoint’s therapy addresses them. There are three main areas of concern with the biologics: 1) the potentially severe side effects, 2) an inconvenient dosing regimen which results in poor patient compliance, and 3) the high cost of the therapies.
The Significant Side Effects of Biologics and How SetPoint’s Therapy Addresses Them
Biologics come in several forms, but all of them have side effects that can be quite severe, most notably a greater susceptibility to serious infections and a higher risk of certain cancers. The increased danger of infection is a direct result of the biologics’ mechanism of action: they suppress the body’s natural immune response. While effective in treating autoimmune diseases, biologics leave patients vulnerable to infections that can be serious, and even life-threatening. In fact, all of the FDA approved biologics for autoimmune diseases carry a “black box” warning about the risks of immunosuppression. SetPoint’s therapy eliminates this shortcoming because, unlike biologics, it does not shut down the body’s immune response. Instead, it uses a highly targeted, neuro-modulatory approach to downregulate the immune system without causing immunosuppression using the body’s innate pathways.
Normally, when the brain detects elevated levels of inflammation in the blood, it sends a signal to the immune system which instructs the white blood cells to “turn off” their inflammatory response. The signal from the brain to the immune system is sent through the vagus nerve, a cranial nerve that starts at the brain stem, travels down both sides of the neck, and then branches out into a network of nerves to innervate all the organ systems in the body. Scientists call this interaction between the brain and the immune system the “inflammatory reflex.” In patients with autoimmune disease, this reflex is not working properly.
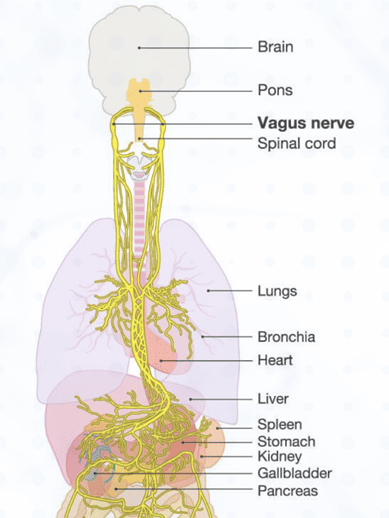
SetPoint’s device is a small neurostimulator which is placed around the left vagus nerve through a narrow incision in the neck in a short outpatient procedure. It works by sending a short electrical signal to the spleen—a major organ involved with the immune system—for one minute, once a day, which may be sufficient to reset the inflammatory reflex to “normal” and thereby reduce the level of inflammation without causing immunosuppression.
“SetPoint’s device works by sending a short electrical signal to the spleen—a major organ involved with the immune system—for one minute, once a day, which may be sufficient to reset the inflammatory reflex to “normal” and thereby reduce the level of inflammation without causing immunosuppression.”
Inconvenient Dosing and Poor Patient Compliance
Biologics are a powerful but non-specific approach to treatment. They, unfortunately, don’t work for all patients, and when they do, they often lose efficacy over time, causing physicians to change therapies for patients frequently throughout the course of their disease. In addition, many of the biologics can only be taken by injection—either by IV infusion (which requires a visit to a doctor’s office every two to four weeks) or by intramuscular (IM) injections (which requires the patients to inject the drug into their stomach and/or thighs every two weeks). Because of these issues, patient compliance with biologic therapies is very poor, with over 50 percent of patients discontinuing therapy within two years. As a result of these challenges only a minority of RA patients achieve sustained remission.
SetPoint’s implantable device eliminates the inconvenience of IV infusion or IM injections and ensures that treatment is administered consistently over the long term.
Most patients with autoimmune diseases are unhappy with their current therapies. Studies have shown that as few as 25 percent of patients treated with biologics are satisfied with their therapies and that many patients are looking for other alternatives. SetPoint’s implantable device eliminates the inconvenience of IV infusion or IM injections and ensures that treatment is administered consistently over the long term. In one patient survey a sizable group of autoimmune patients said they would prefer the SetPoint option even before trying a biologic.

High Cost of Biologics
Biologics are very expensive therapies, typically ranging from $1,300 to $3,000 a month, to as high as $5,000 a week. As a result, spending on autoimmune diseases reached $77 billion in the U.S. in 2020—more than what was spent on cancer or diabetes. By offering a long-term treatment that can be provided in a single outpatient procedure with the possibility of reducing the need for biologic therapies, SetPoint’s therapy has the potential to meaningfully reduce the cost of care for autoimmune diseases.
“SetPoint’s therapy has the potential to meaningfully reduce the cost of care for autoimmune diseases.”
SetPoint Medical has an impressive leadership team, headed by CEO Murthy Simhambhatla, PhD, and Chief Medical Officer David Chernoff, MD. The SetPoint system has been granted Breakthrough Device Designation by the FDA for patients with RA who have an incomplete response to, or are intolerant to, biologic drugs.

The company is currently enrolling the second stage of its pivotal trial which began last fall, after the FDA completed an interim review of the results from the first 60 patients that were enrolled in the study and gave the company permission to proceed with the trial. The company expects to announce the results from the pivotal trial in mid-2024 and to submit its application for FDA approval by the end of that year.
As part of the recent financing, Zack Scott, MD, a general partner on Norwest’s healthcare venture capital team, has joined the board of directors at SetPoint. Norwest is excited by the prospects for SetPoint Medical as a company, as well as the potential to transform the lives of millions of people who suffer from autoimmune diseases, and we look forward to working with the company and its other investors to bring SetPoint’s exciting new therapy for autoimmune diseases to market.